
By David Donaldson, B.Agr, N.Sch, P.A.S.
Molybdenum is required in small amounts in the diets of farm animals.
No discussion on the requirements of Molybdenum (Mo) can be considered without also considering the Copper requirements of the animal. Early interest in Molybdenum (Mo) was less concerned with the daily requirement for Mo than the effects of high concentrations, which is known to reduce Copper absorption (see Trace Mineral Interactions).
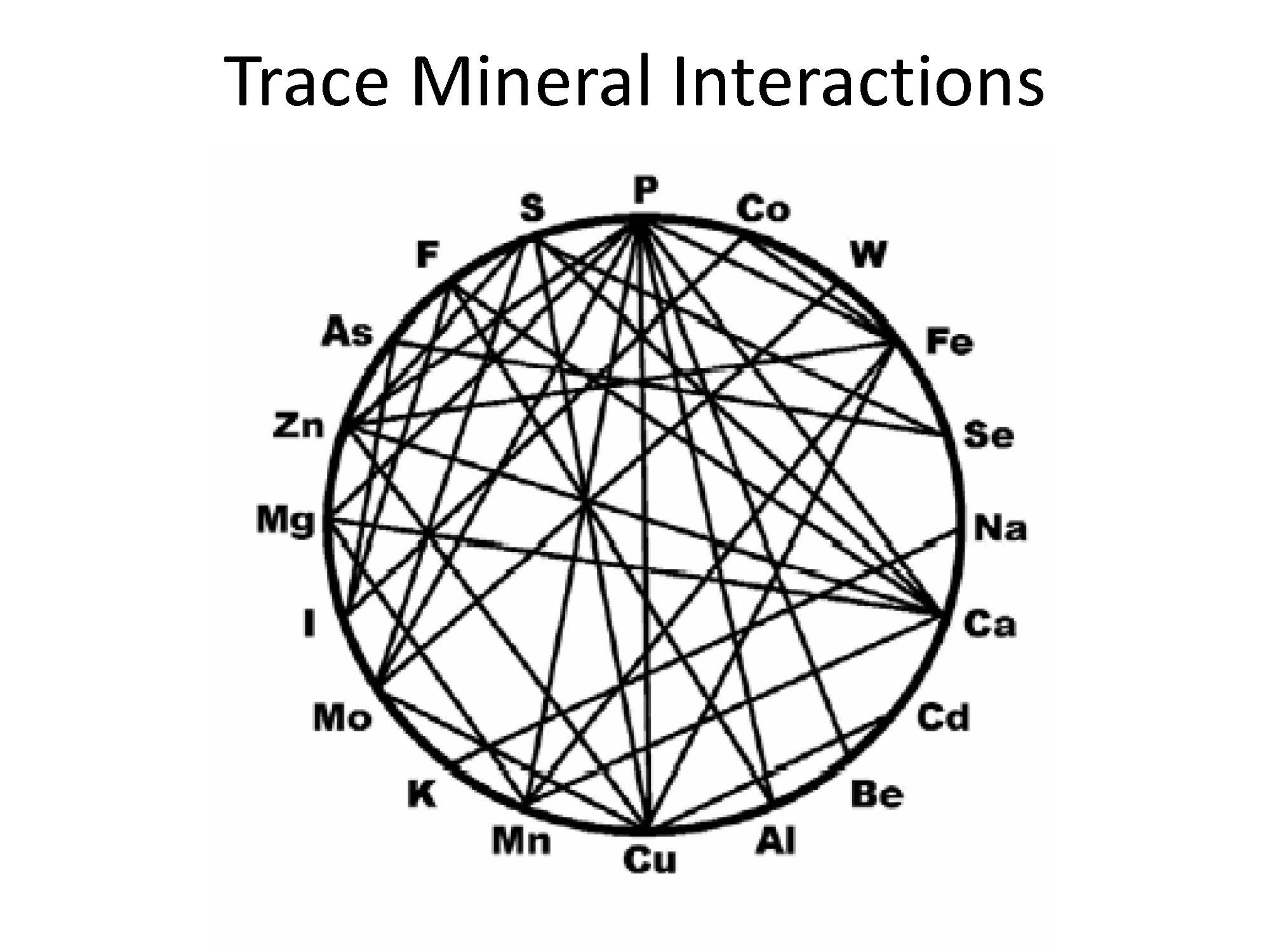
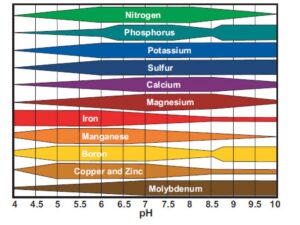
Mineral Concentration in Soil Based on pH
Soils vary widely in their Mo content and the subsequent uptake by forage crops. Maize (Corn) silage tends to have the lowest concentrations, grass and alfalfa are intermediate and pasture is the most variable. Concentrations of over 60ppm (parts per million) have been measured in the pasture (see Mineral Concentration in Soil Based on pH).
Soil concentrations of Mo can range from 0.1ppm to over 20ppm. Sandy soils tend to hold the least. Typically around 10% of Mo is available to the plant, but this amount rises as the pH of the soil increases. Poorly drained soils, peat and organic-rich soils, alkaline soils and liming all increase Mo uptake and reduce Sulfate uptake. Agronomists are often aware of soils that are low in Mo, as crop yields will respond to additional Mo in these situations.
Plants and grains typically range from 0.5 to 2.5ppm Mo in the DM, so Mo concentrations are almost always sufficient to meet the requirements of the animal. At concentrations above 2ppm DM, Mo can form an insoluble complex with Sulfur in the rumen. This complex binds with Copper, thereby reducing Copper availability.
It is rarely necessary to supplement Mo in diets, but in ruminants, on high fibre diets with high Copper:Mo ratios (greater than 4:1), most of Mo can be excreted in the manure. The only element known to tie up Mo is Tungsten (chemical symbol W). Tungsten concentrations in soil are typically very low but are known to accumulate around surface mining sites.
Cattle are the least tolerant of farm livestock to high concentrations of Mo, closely followed by sheep. Pigs and poultry can tolerate levels more than 10 times higher than cattle. When cattle are placed on High Mo pastures (over 20 ppm), they quickly develop severe scours.
Mo is required in a few enzyme systems, principally Xanthine oxidase (XO), Sulfide oxidase (SO) and Aldehyde oxidase (AO). The first indication of an essential role for Mo came from the discovery that the enzyme XO contained Molybdenum and that its activity depended on the presence of Mo (de Renzo et al,1953; Richert and Westerfield, 1953).
These enzymes are found in Milk and many tissues (Mills and Davis, 1987). Mo, as a co-factor in these enzymes, aids in catalyzing the oxidation or metabolism of sulfur-containing amino acids, purines, pyrimidines, and aldehydes. In poultry, Mo is involved in the formation of Uric acid, which is their method of excreting excess protein.
Absorbed Mo is stored in tissues as molybdopterin, bound to XDH and AO in the cytosol and to SO in mitochondrial membranes.
Nonruminant animals and milk-fed calves metabolize Mo quite differently than ruminants. In single stomached animals, Mo is absorbed very efficiently across the stomach (van Campen and Mitchell 1965) and abomasum wall (Miller et al 1972). Absorption is via an active system (energy-dependent), shared with and inhibited by Sulfate. Since very little Sulfur leaves the rumen as Sulfate, this antagonism is only a feature of non-ruminants.
Excess Mo is excreted in the urine. In the renal tubule, sulfate prevents resorption of Mo and increases Mo excretion (Bishara and Bray, 1978). Both non-ruminants animals and ruminants demonstrate this same antagonism in the kidneys.
Deficiency
Very rare in all animal species and is likely as a result of high Copper and Sulphur. Symptoms include:
Toxicity
NRC quotes a maximum of 10ppm in the dry matter for cattle, but even at much lower levels, Copper availability will be affected. Pigs and poultry can tolerate much higher concentrations. Toxicosis symptoms are essentially the same as a Copper deficiency, namely;
Requirements
The precise requirement for Mo is hard to define because of the relationship to the Copper and Sulphur content of the diet. A dietary dry matter concentration of 0.5ppm is sufficient for all species except when Sulphur and Copper levels are higher than normal. AK