
By Dr. David Jones, Ph.D., P.A.S.
Chromium is an important trace mineral. In fact, evidence for an essential role for chromium in humans came from patients receiving total parenteral nutrition (Anderson, R.A. 1995. Chromium and parenteral nutrition. Nutrition. 11:83-86). Patients developed diabetic symptoms that were alleviated by the addition of chromium. This finding created interest in chromium as a dietary supplement.
Chromium is part of what is known as the glucose tolerance factor. Glucose tolerance factor is involved with the effect of insulin on tissues, either by stabilizing the insulin molecule or by facilitating the interaction of insulin with its receptor in tissues. The glucose tolerance factor is inactive without chromium. This is an important mechanism concerning the metabolism of glucose for energy. Studies suggest that chromium affects insulin by binding to chromodulin (low molecular weight chromium binding substance), a molecule that amplifies insulin receptor activity (Diagram 1). When insulin concentrations decrease, chromodulin is thought to be released from the cells and excreted in the urine.
Diagram 1. A proposed model for the enhancing effects of chromium on insulin activity
*LMWCr = low-molecular-weight chromium-binding substance
Adapted from Vincent, J.B. Quest for the molecular mechanism of chromium action and its relationship to diabetes. Nutr Rev. 2000; 58:67-72. (as found online from the Linus Pauling Institute; Oregon State University).
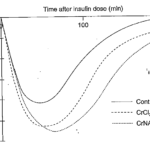
Insulin is secreted by specialized cells in the pancreas in response to increased blood glucose concentrations. Insulin binds to insulin receptors on the surface of cells, which activates the receptors and stimulates glucose uptake by the cells. This provides the cells with glucose for energy and prevents blood glucose from becoming elevated. Research with calves has shown an intensified response to insulin with the addition of chromium given as chromic chloride or chromium-nicotinic acid complex (figure 1., Kegley et al., 1997). Insulin also influences the metabolism of fat and protein.
Figure 1. Continuous supplements of chromic chloride (CrCl3 and chromium-nicotinic acid complex (CrNA)
Although chromium comes in many forms/states, chromium occurs primarily in its trivalent (Cr+3) or hexavalent (Cr+6) form. The principal component of chromium ores is chromite, a black mineral consisting largely of iron and Cr+3 oxides. Approximately 60% of the chromium processed is used in the production of stainless steel and other alloys.
Dietary chromium is primarily absorbed from the small intestine. Inorganic forms of chromium are very poorly absorbed which has led to their use as internal markers for digestibility studies. Complexing chromium with organic compounds (e.g. Chromium Propionate) has increased the absorption of chromium when fed. Chromium from naturally occurring sources (e.g. brewer’s yeast) also is efficiently absorbed.
The role of chromium in animal nutrition has demonstrated the following results:
The responses to supplemental chromium seemed to be greater in animals that are under some physiological stress. Cortisol is a hormone produced during stress. Cortisol is known to negatively regulate immune function during stressful times. A recent study (Burdick et al., 2011) found that chromium propionate mitigated the immune response by reducing proinflammatory cytokines in stressed cattle.
It has been recommended that chromium be supplemented at 0.5 ppm in the ration for transition cows (3 weeks before calving to 4 weeks after calving). Only Chromium Propionate is allowed for use in the U.S. about ruminant diets. Both Chromium picolinate and chromium propionate can be fed to swine. There is the potential for 3 or more pounds of milk when Chromium is fed during the transition period. The cost of chromium may prevent a return on investment if it is fed for more than the transition period.
Chromium+3 absorption is increased by ascorbic acid (vitamin C) and niacin (Vitamin B3). Various amino acids (e.g. tryptophan) can be converted to precursors (e.g. picolinate) of bioactive forms of chromium.
Chromium is transported through the blood by the iron transport protein transferrin. Therefore, chromium has a negative interaction with iron as these two trace minerals compete for one of the binding sites on the transferrin protein. Iron overload can saturate transferrin with iron which may interfere with chromium transport by leaving fewer available binding sites on transferrin for chromium. This can lead to a chromium deficiency, should it occur. Reducing the chromium available to the cells can lead to cellular insensitivity to insulin and a diabetes type syndrome. Water and feed high in iron need to be monitored, as they can potentially lead to a reduction in chromium absorption and transport to the cells.
Diets high in refined or simple sugars increase urinary chromium losses while diets that produce propionate in the rumen (glucose precursor) would be expected to deplete body chromium stores, increasing the requirement for chromium. Supplemental fat may induce insulin resistance or impair glucose tolerance increasing chromium requirements.
Clinical signs of chromium deficiency include:
Trivalent chromium (Cr+3) is safe to add to diets. Chromium toxicity is primarily associated with hexavalent chromium (Cr+6) as it is approximately 5 times more toxic than Cr+3. Hexavalent chromium passes into the interior of cells more readily than Cr+3, depressing mitochondrial oxygen consumption. If significant amounts of Cr+6 reach the cell nucleus, there can be pathological changes in the DNA. Fortunately, it is unlikely that significant amounts of Cr+6 will be consumed orally or absorbed because it is reduced in the environment and the stomach to the more stable, and safe, Cr+3. Insoluble forms of Cr+3, such as chromic oxide, are very poorly absorbed. Concentrations of insoluble Cr have been fed as high as 3000 mg/kg of the diet with no adverse effects. Soluble forms of Cr+3 can be fed as high as 1000 mg/kg of diet for ruminants and 500 mg/kg of diet for poultry.
Signs of chromium toxicity include:
To learn more about trace minerals, dissect The Advantage for more articles.
References
Anderson, R.A. 1995. Chromium and parenteral nutrition. Nutrition. 11:83-86
Burdick, N.C., B.C. Bernhard, J.A. Carroll, R.J. Rathmann, and B.J. Johnson. 2011. Enhancement of the acute phase response to a lipopolysaccharide challenge in steers supplemented with chromium. Innate Immun. Published early online Dec. 16.
Kegley, E.B., Spears, J.W. and Eisemann, J.H. 1997. Effect of shipping and chromium supplementation on performance, immune response and disease resistance of steers. Journal of Animal Science. 75: 1956-1964.
Mowat, D.N. Supplemental organic chromium for beef and dairy cattle.
Mineral Tolerance of Animals. 2005. Second revised ed. p. 115-123.
Nutrient Requirements of Dairy Cattle. 2001. Seventh revised ed. p. 146-147.
Nutrient Requirements of Beef Cattle. 1996. Seventh revised ed. p. 61.
The Mineral Nutrition of Livestock. 2001. Third ed. p. 521.